- Blog
- Battlefield 2042 servers
- End of drug lord business zombie night terror
- Hot door cadtools 12-2-6 for adobe illustrator
- Mr crab 2 apk
- Suit up simple desktops
- Syberia ii origin on the house
- Garmin express won t install
- If it all right i want to kick it with you all night
- Gesundheit institute
- Calcium atomic radius
- -sweet home 3d house
Ingesting or inhaling calcium metal can be fatal due to the burns it can produce.
#Calcium atomic radius skin#
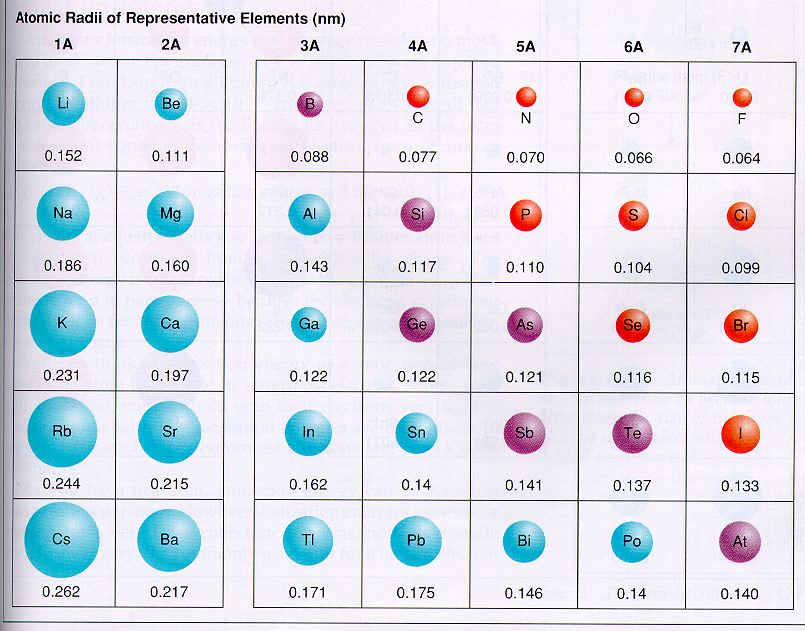
Skin contact with calcium metal can cause irritation, corrosion, and chemical burns. Calcium metal reacts exothermically with water or acid.People report is as contributing a mineral, sour, or salty flavor. People and other animals can often taste the calcium ion. However, even for atoms of the same type, atomic radii can differ, depending on the oxidation state, the type of bonding.Calcium metal is soft enough to cut with a knife, although somewhat harder than the metal lead.It is the fifth most abundant element on earth. It is a soft, silvery-white alkaline earth metal, whose name comes from the Latin word calx, which means lime. Calcium salts are used to produce orange in fireworks. Calcium (Ca) is a chemical element of the periodic table, located in the group 2 and the period 4, and is having the atomic number 20. Calcium is used in fireworks to deepen the color.For FCC structure ‘a’ and radius of atom ‘r’ related as follows. Where a is the length of the edge of the cube. To calculate the density we need to know the mass of 4 atoms and volume of 4 atoms in FCC unit cell. It must be noted, atoms lack a well-defined outer boundary. That means one unit cell contains total 4 calcium atoms. Calcium burns with a dark red color in a flame test. The atomic radius of Calcium atom is 176pm (covalent radius). Calcium is a chemical element with symbol Ca and atomic number 20.Approximately one third of the mass of the human body is calcium after all water is removed. Calcium is the fifth most abundant element in the human body.

However, too much calcium can lead to kidney stones or artery calcification.
#Calcium atomic radius free#
Calcium is not found free in nature, but calcium compounds are common. On moving down the group, the atomic radii increase as additional electron shells are added.Calcium is the 5th most abundant element in the Earth's crust, making up 3.22% of the earth, air, and oceans.
